Exploring the mechanism of dihydromyricetin in improving renal fibrosis in db/db mice based on network pharmacology and experimental verification
Diabetes nephropathy (DKD) is one of the major microvascular complications of type 2 diabetes mellitus (T2DM) and the main cause of end-stage renal disease. Its main characteristics are thickening of the glomerular basement membrane, expansion of the mesentery and tubulointerstitial matrix, and loss of podocytes, leading to the development of microalbuminuria and decreased renal function. With the continuous development of diabetic kidney disease (DKD), the microstructure of renal tissue undergoes persistent inflammation and damage, leading to diffuse interstitial fibrosis that destroys the normal renal microstructure and ultimately develops into renal fibrosis (RF). Currently, the means to alleviate the progression of DKD to RF mainly focus on improving DKD. However, the mechanism of most complications of diabetes, including DKD, has not yet been clarified, so that the complex mechanism in the process of DKD evolving into RF is not clear, leading to unsatisfactory therapeutic effects of DKD and RF. Therefore, it is urgent to explore the pathogenesis of DKD and discover new therapeutic targets to inhibit its development.
Dihydromyricetin (DHM) is a natural flavonoid compound with various pharmacological effects, including anti-inflammatory, antioxidant, free radical scavenging, improvement of mitochondrial dysfunction, and regulation of autophagy. At present, multiple studies have shown that DHM has great potential in the treatment of metabolic diseases. It can not only improve insulin resistance and lower blood glucose levels by inducing autophagy, improving oxidative stress, inhibiting inflammation, and increasing mitochondrial activity, but also; At the same time, it can improve many complications of diabetes, such as diabetes encephalopathy, diabetes cardiomyopathy, etc. However, the research on DHM improving the complications of diabetes is not sufficient, and the related pharmacological mechanism still needs to be further explored. In addition, there is limited research on whether DHM can further improve the RF caused by DHM, and the therapeutic effect is still unclear.
Network pharmacology can construct a “drug component target disease” interaction network based on the structure of traditional Chinese medicine components, their biological effects, and disease-related targets in the context of a vast network of biological processes. It is an effective means of exploring the effective components, molecular mechanisms, and potential targets of traditional Chinese medicine. Molecular docking can simulate the interaction between molecules and proteins, predict the conformation of ligands and receptors, calculate and predict affinity and binding modes to verify the specific mode of action of drug components on major targets. Therefore, this study used network pharmacology to screen and predict potential targets of DHM on DKD and RF, constructed a “component target disease” interaction network, and validated the predicted main targets through molecular docking and in vivo experiments. The molecular mechanism of dihydromyricetin improving renal fibrosis in db/db mice was explored, providing a theoretical basis for the clinical application of DHM.



At present, the common treatment of diabetes mainly depends on the control of blood sugar, while DKD, one of the most serious complications of diabetes, has no effective treatment strategy. Therefore, it is very important to have a deep understanding of the pathogenesis of DKD and to find effective treatment strategies to suppress the trend of DKD developing into renal fibrosis. Natural medicines have been proven to have unique advantages of fewer side effects and multi-target efficacy, making them an important source for the development of new drugs for chronic diseases in recent years. Further research on DHM has revealed significant therapeutic potential in improving metabolic diseases. Therefore, based on the improvement of renal fibrosis in db/db mice by DHM, this study combines network pharmacology and molecular docking technology to explore the potential therapeutic targets and molecular mechanisms of DHM in improving renal fibrosis in db/db mice, and verifies them through animal experiments.
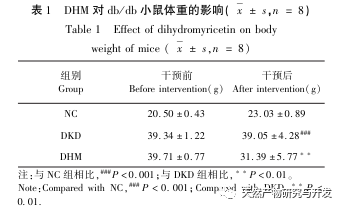
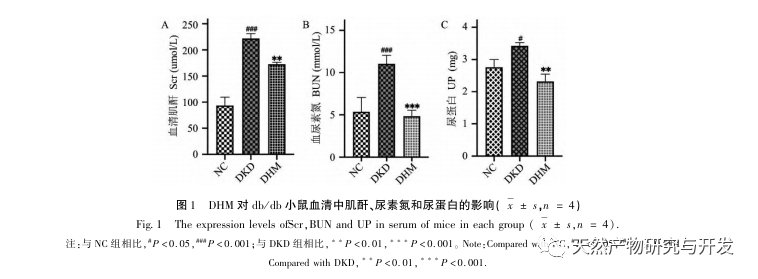
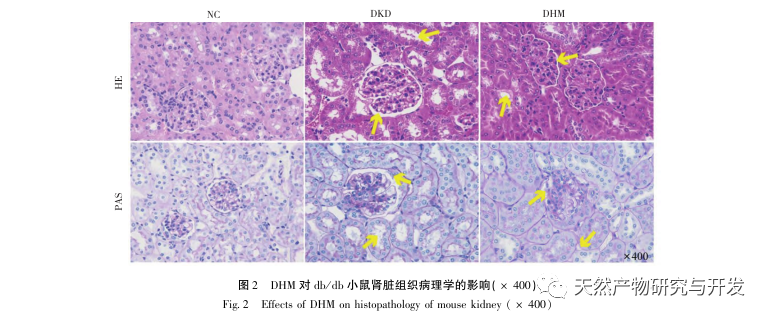
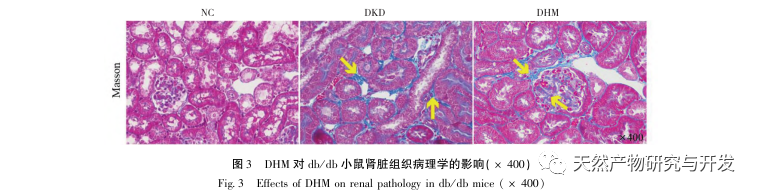
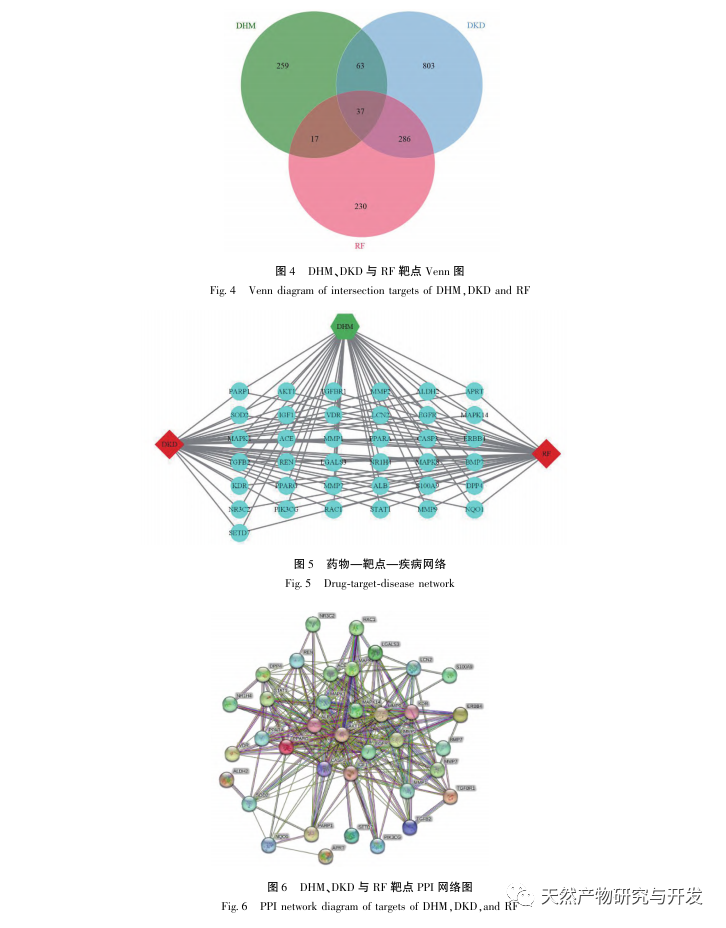
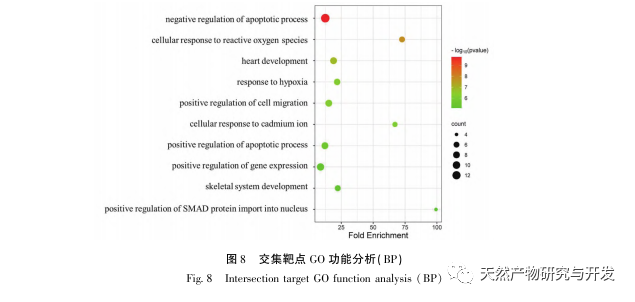
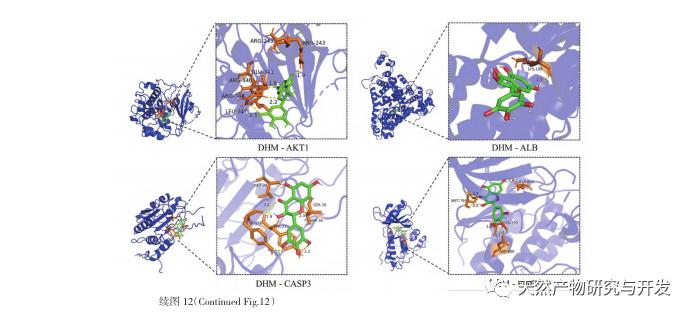
Firstly, we observed that 10 weeks of DHM intervention can effectively alleviate blood glucose, body weight, creatinine, urea nitrogen, urinary protein, and renal pathological manifestations in db/db mice, indicating that DHM can alleviate renal function damage and slow down the development of DKD in db/db mice. After analyzing and screening 37 intersecting targets of DHM, DKD, and RF, it was found that 10 targets, including AKT1, ALB, CASP3, EGFR, MMP9, IGF1, PPARG, KDR, MMP2, ACE, may be key targets for DHM to improve DKD. AKT1, CASP3, EGFR, MMP9, IGF1, MMP2, ACE have been confirmed to be activated in the environment of diabetes, and specific inhibition can improve the damaged state of DKD and slow down the development trend of fibrosis. PPARG and KDR are inhibited in the physiological environment of diabetes, and play a role in renal protection after activation. ALB is an important biochemical indicator to observe the degree of kidney damage during the development of diabetes. It usually increases with the occurrence of DKD and decreases after improvement of DKD. This means that the 10 key genes we have screened can not only serve as potential biomarkers for DKD, but also as key targets for further exploring the pathogenesis and treatment of DKD.
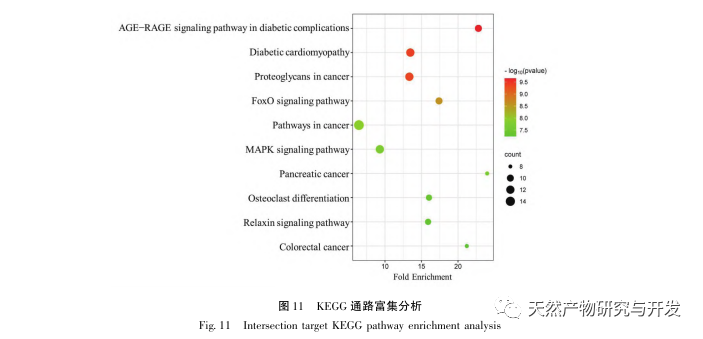
KEGG pathway enrichment analysis showed that the improvement of DHM on DKD might involve AGE-RAGE signaling pathway, diabetes cardiomyopathy and FoxO signaling pathway. The AGE-RAGE signaling pathway and FoxO signaling pathway play key regulatory roles in the development of DKD. Research has found that AGEs are specifically activated in endothelial cells, renal tubules, mesangial cells, and podocytes of DKD patients, and their interaction with RAGE is enhanced, further exacerbating oxidative stress and inflammation in the kidneys, leading to the deterioration of DKD. In addition, activated FoxO3a can inhibit the level of oxidative stress in the kidney of diabetes rats, improve inflammation, and then inhibit the occurrence of DKD. Indicating that specific regulation of the AGE-RAGE signaling pathway or FoxO signaling pathway may be of great significance for the prevention and treatment of DKD.
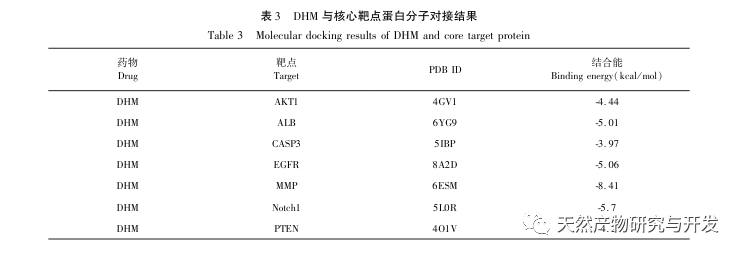
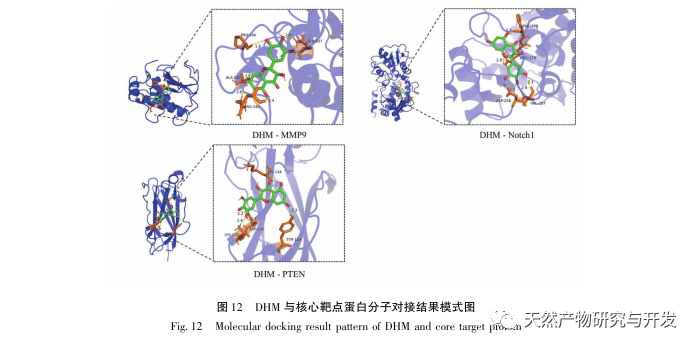
The molecular docking results showed that DHM exhibited good binding affinity with the top 5 selected targets, indicating that the effectiveness of DHM in treating DKD has been validated at the molecular level. In addition, we noticed that the key target gene AKT1, which ranked first in the screening, was closely related to the AGE-RAGE signaling pathway and FoxO signaling pathway in the development of DKD through enrichment analysis of the KEGG pathway. The AGE-RAGE signaling pathway can synergize with the PI3K/Akt signaling pathway to induce oxidative stress and chronic inflammation in the kidneys, leading to kidney disease. The activation of PI3K/Akt/FoxO3a pathway can improve the physiological function of kidney in diabetes. Therefore, we further attempted to investigate the interaction between the Notch pathway upstream of AKT1 and DHM and conducted molecular docking validation. The molecular docking results showed that DHM exhibited good binding affinity with Notch1 and PTEN.
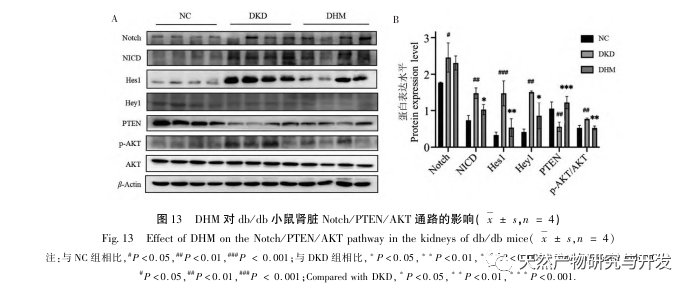
Previous studies have shown that the Notch pathway is overactivated during the development of DKD, manifested by increased expression of Notch1, NICD, Hes1, and Hey1. Inhibitors of Notch1 can activate PTEN by reducing the activation of Hes1, thereby improving autophagy disorders and slowing down the occurrence of RF. In addition, other studies have found that PTEN activation can also alleviate the progression of DKD by inhibiting the PI3K/Akt pathway. Based on the above results, we speculate that DHM can improve DKD by regulating the Notch/PTEN/AKT pathway. Western blot results showed an abnormal increase in the activation of the Notch pathway, a decrease in PTEN expression, and an increase in AKT phosphorylation in the kidneys of db/db mice. And DHM intake can inhibit the activation of Notch pathway, activate PTEN, and inhibit the phosphorylation of AKT. Although the Notch/PTEN/AKT signal axis as a whole pathway has rarely been studied on renal protection in diabetes, our research has improved relevant research to a certain extent, and enriched the molecular mechanism of DHM’s biological protection through multiple targets.
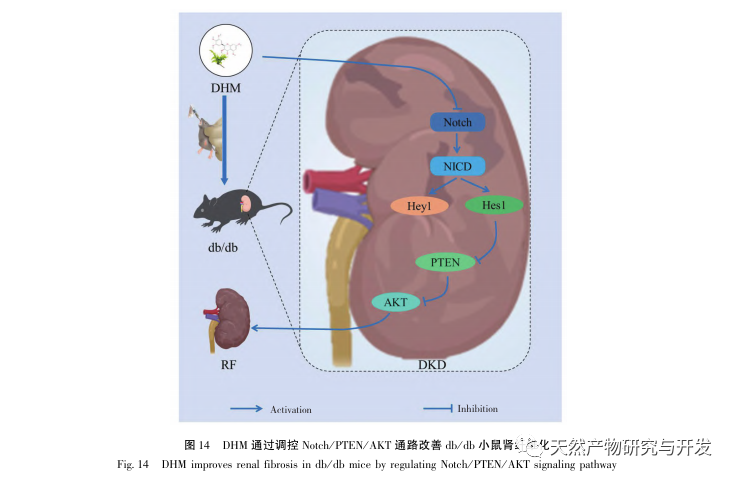
In summary, based on the observation of DHM improving the pathological manifestations of DKD, this study combined network pharmacology and molecular docking technology to preliminarily demonstrate that the role of DHM in improving the progression of DKD may be achieved through key targets such as AKT1, ALB, CASP3, EGFR, MMP9, IGF1, PPARG, KDR, MMP2, ACE, and analyzed the relevant pathways; Finally, preliminary validation and analysis were conducted on the potential AKT targets of DHM through animal experiments, and it was found that DHM may play a role in preventing and treating DKD by regulating the Notch/PTEN/AKT pathway (see Figure 14). However, there is currently relatively little research on the complications of DHM in T2DM, and its therapeutic potential still needs further investigation.
