Flavonoids from the outer skin of sour soybean and their antioxidant activity evaluation
Tamarinus indica Linn. is a leguminous plant, whose fruit is also known as Luo Huang Zi, Sour Horn, Sweet Horn, Luo Wang Zi, etc. It is native to Africa and is now cultivated in various tropical regions. Commonly found in Taiwan, Fujian, Guangdong, Guangxi, southern, central, and northern Yunnan (Jinsha River Valley). Sour beans have a long history of medicinal use. The “Compendium of Materia Medica” records that “Luo Huang Zi… treats the growth of insects in the organs and the abdominal pain caused by children eating soil”; The “Dian Nan Ben Cao” records that “sour dumplings… treat the transformation of wine into phlegm and isolate it in the stomach. Sour beans contain different structural types of substances such as flavonoids, anthocyanins, oligomeric anthocyanins, terpenes, polysaccharides, and organic acids, which have biological activities such as antioxidant, anti-inflammatory, anti-aging, and blood pressure lowering.
In recent years, there has been an increasing amount of research on the flowers, leaves, fruits, and seeds of sour beans both domestically and internationally. However, there are currently few reports on the composition and activity of the outer skin of sour beans. In order to clarify the composition and antioxidant activity of the outer skin of sour beans, this experiment studied the chemical composition and antioxidant activity of acetone extract from the outer skin of sour beans, in order to provide a scientific basis for the subsequent medicinal and development of sour beans.
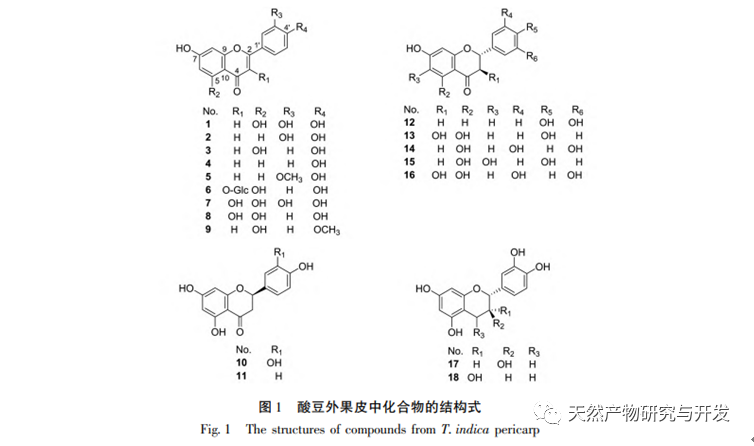
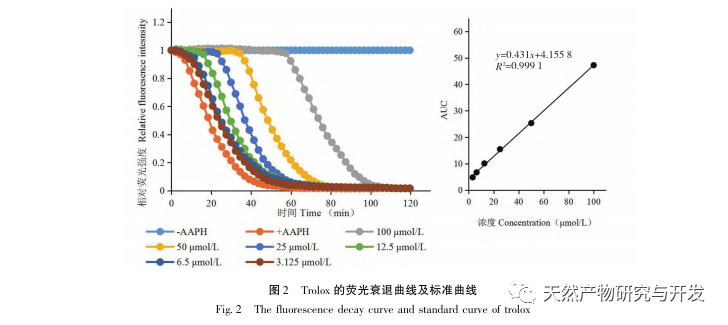
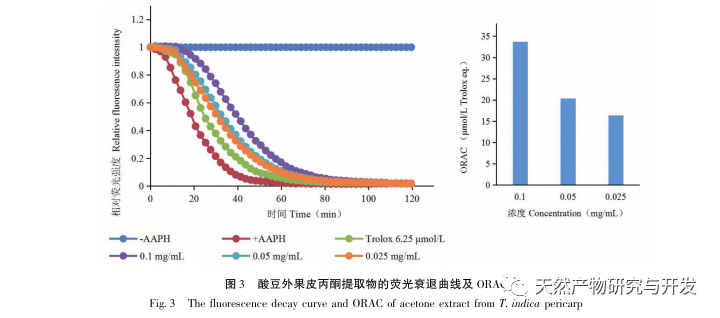
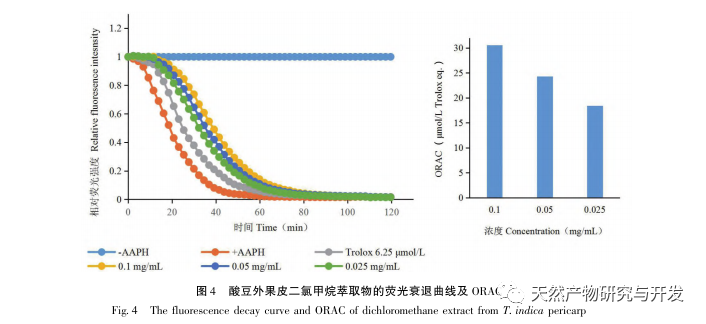
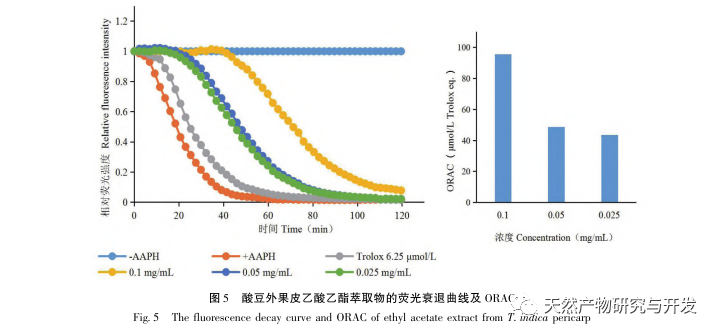
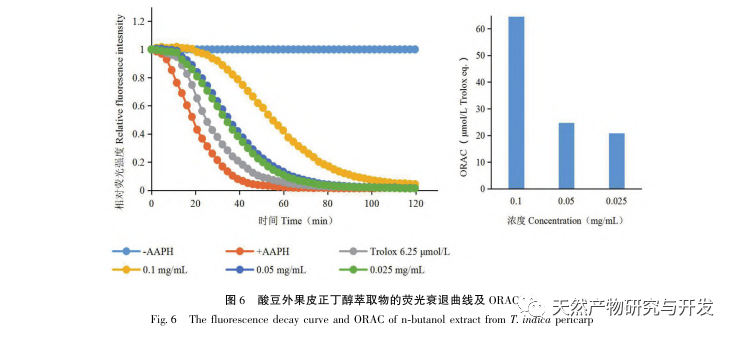
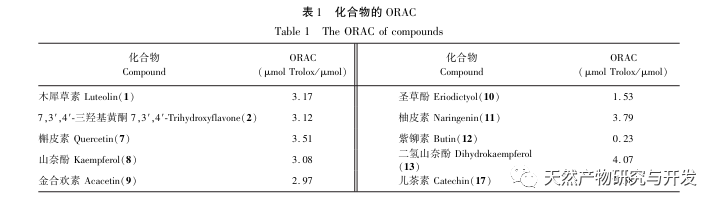
Sour beans contain different structural types of substances such as flavonoids, organic acids, anthocyanins, oligomeric anthocyanins, polysaccharides, and terpenes. However, the outer skin of sour beans is often overlooked as a byproduct of food processing. In this experiment, a total of 18 flavonoid compounds were isolated from the outer skin of sour beans, including 6 flavonoids (1-5, 9), 2 flavonols (7, 8), 5 dihydroflavonoids (10-12, 14, 15), 2 dihydroflavonols (13, 16), 2 flavanols (17, 18), and one flavonoid glycoside (6). Among them, 10 compounds (2, 4-6, 9, 11, 13-16) were isolated from this genus of plants for the first time, indicating that the outer skin of sour beans contains a large amount of flavonoids, which is consistent with the large amount of components contained in sour beans. The ORAC experiment results showed that the acetone extract, dichloromethane extract, ethyl acetate extract, n-butanol extract, and 9 compounds (1, 2, 7-10, 12, 13, 17) from the outer skin of sour beans all had good in vitro antioxidant activity.
The antioxidant activity of flavonoids is closely related to their structure, and the number and position of hydroxyl groups, as well as the presence of carbonyl groups in the C-ring, all have a certain impact on the antioxidant activity of flavonoids. Due to the direct involvement of phenolic hydroxyl groups in free radical scavenging, the amount of substitution of phenolic hydroxyl groups plays an important role in the antioxidant effect of flavonoids. The experimental results showed that the ORAC value of dihydrokaempferol in the dihydroflavonoid type was higher than that of naringenin, and quercetin was higher than other flavonoid types such as luteolin and kaempferol. It can be seen that the more hydroxyl groups there are, the stronger the antioxidant capacity of the compound. Research has shown that the main site of antioxidant activity of flavonoids is the substitution of hydroxyl groups in the B ring, with the B-ring adjacent phenolic hydroxyl group having the strongest activity. This phenomenon may be due to the fact that the adjacent phenolic hydroxyl group on the B ring is more susceptible to electron attack. Experimental results show that the number of hydroxyl groups in magnolol and kaempferol is the same, but the ORAC value of magnolol is higher. It is speculated that the B-ring hydroxyl group may have more antioxidant activity than the A-ring hydroxyl group, and its adjacent phenolic hydroxyl group plays a significant role; Double bonds are electron withdrawing groups, and their induction effect can reduce the antioxidant capacity of flavonoids. The presence of double bonds can also prolong the conjugated system of flavonoids, enhance the stability of free radical products, and improve antioxidant activity; The experimental results showed that when the substituents are the same, the ORAC value of dihydrokaempferol is greater than that of kaempferol, while the ORAC value of purpurin is greater than that of 7,3 ‘, 4’ – trihydroxyflavone. It can be seen that the influence of the presence of C-ring double bonds on its antioxidant activity still needs to be studied; The ORAC value of dihydrokaempferol is significantly higher than that of catechins, indicating that the presence of the 4-carbonyl group has a certain promoting effect on antioxidant activity. This experiment analyzed the antioxidant effects of some flavonoids and the relationship between their structures, and verified the relevant conclusions on the impact of the above structures on the strength of antioxidant capacity.
